
How LIMS Is Solving the Compliance Crisis Before It Starts
Table of Contents
- Introduction — The Pressure on Water Testing Labs
- What Is LIMS and Why Does It Matter?
- The Compliance Landscape in 2026
- How Compliance Failures Actually Happen
- Core LIMS Features Every Water Lab Needs
- Sample Lifecycle Management
- Quality Control — Built Into the Workflow
- Regulatory Reporting Made Simple
- Chain of Custody — The Audit Trail That Protects You
- LIMS vs. Manual Systems — A Practical Comparison
- Choosing the Right LIMS for Your Water Lab
- Implementation — What the Journey Looks Like
- ROI — The Business Case for LIMS
- What a Purpose-Built Water Lab LIMS Looks Like in Practice
- Conclusion & Next Steps
1. Introduction — The Pressure on Water Testing Labs Labs
Running a water testing laboratory in 2026 is a different job than it was even five years ago. The volume of samples has grown. The list of parameters to test has expanded. Regulatory bodies are stricter, deadlines are shorter, and clients expect digital reports delivered in real time — not mailed a week later.
And somewhere in the middle of all that pressure, a missed signature on a chain-of-custody form, a result filed in the wrong column, or a sample that sat past its hold time can spiral into a compliance crisis that damages client relationships, triggers regulatory action, and in the worst cases, leads to the suspension of lab accreditation.
This guide is for the lab managers, quality officers, and technical directors who are already feeling that pressure — and who are looking for a structured, practical answer.
That answer is a Laboratory Information Management System — LIMS. But not all LIMS platforms are created equal, and a water testing environment has unique requirements that generic platforms often overlook.
What follows is a comprehensive walkthrough of how a purpose-built LIMS transforms the way water testing labs operate — from the moment a sample arrives at the reception desk to the moment a final report lands with a regulator or a client.
Key Insight
The labs that are navigating 2026 confidently are not necessarily the biggest labs. They are the ones with the best operational systems — and LIMS is at the center of those systems.
Throughout this document, we draw on real-world laboratory workflows, regulatory requirements across global markets, and the experience of water testing labs that have made the transition from manual processes to modern LIMS platforms.
2. What Is LIMS and Why Does It Matter?
A Laboratory Information Management System — LIMS — is a software designed to manage the complete lifecycle of laboratory data and samples. The definition sounds straightforward, but the implications for a water testing lab are profound.
A well-implemented LIMS is not just a database. It is the operational backbone of the entire laboratory — the system that connects people, instruments, samples, methods, and reports into a single, traceable workflow.
What LIMS Actually Does
At its core, a LIMS manages:
- Sample registration and login — capturing every incoming sample with a unique identifier, collection details, client information, and required tests
- Test assignment and scheduling — directing each sample to the right analyst, instrument, or department based on the test parameters requested
- Data capture and verification — storing test results, applying quality control checks, and flagging anomalies before they reach the final report
- Reporting and distribution — generating compliant, formatted reports and delivering them to clients or regulatory bodies
- Audit trail maintenance — recording every action taken on every sample, with timestamps and user logs, so nothing is ever lost or undisputed
Why Manual Systems Fall Short
Many water testing labs still operate with a combination of spreadsheets, paper logbooks, and email chains. This approach has one significant advantage: it is familiar. It has several significant disadvantages that grow more dangerous as the lab scales.
| Manual System | LIMS-Driven System |
|---|---|
| Data entered manually — human error risk at every step | Data captured automatically from instruments and workflows |
| No real-time alerts for out-of-spec results | Instant alerts when QC thresholds are breached |
| Audit trails reconstructed from paper and email | Full digital audit trail — automatic and tamper-evident |
| Reports created manually — formatting inconsistencies | Compliant, templated reports generated in minutes |
| Compliance checked retrospectively — after the fact | Compliance built into every workflow step |
The table above captures the core difference. Manual systems are reactive — they catch problems after they happen, often too late. A LIMS is proactive — it prevents problems from happening in the first place.
3. The Compliance Landscape in 2026
Water quality testing is one of the most regulated activities in any analytical laboratory environment. The outputs of water labs inform decisions about public health, industrial discharge, environmental protection, and food safety — which is why global regulatory frameworks around water testing have grown increasingly demanding over the past decade.
In 2026, labs are navigating a multi-layered compliance environment that includes international accreditation standards, national regulatory requirements, and client-specific testing protocols — often simultaneously.
Key Regulatory Frameworks
ISO/IEC 17025 — The Baseline for Lab Competence
ISO 17025 is the international standard for testing and calibration laboratories. Any water testing lab that wants to be recognized as technically competent — and most institutional and regulatory clients now require this — must demonstrate conformance with ISO 17025.
Key requirements under ISO 17025 include documented procedures for every test method, full traceability for all measurements, regular equipment calibration, defined QC protocols, and a comprehensive record-keeping system. A LIMS addresses all of these requirements structurally — not as an add-on, but as a function of how the system works.
Beyond ISO 17025, water testing labs must comply with national standards that specify permissible limits for various contaminants, approved testing methods, reporting formats, and submission timelines.
- In India: BIS standards (IS 10500) govern drinking water quality, while CPCB guidelines cover industrial and environmental discharge testing
- In the United States: EPA methods under the Safe Drinking Water Act define approved testing procedures for public water systems
- In the European Union: The Drinking Water Directive sets strict quality parameters and mandates documentation for all monitoring activities
- In the Middle East and Southeast Asia: Regional standards are increasingly aligned with WHO guidelines, with additional national requirements layered on top
Regulatory Trend in 2026
Across all major markets, regulators are moving toward electronic submission of test data — which means labs that cannot generate structured digital reports are at a competitive and compliance disadvantage.
The Accreditation Pressure
Beyond meeting minimum regulatory requirements, many water testing labs are under pressure to maintain or achieve NABL (National Accreditation Board for Testing and Calibration Laboratories) accreditation in India, or ILAC-recognized accreditation in international markets.
Accreditation assessors look at the same things every time: can the lab demonstrate that its data is reliable, traceable, and produced by a documented, controlled process? A lab running on spreadsheets and paper will always struggle to answer that question convincingly.
4. How Compliance Failures Actually Happen
Most compliance failures in water testing labs are not the result of deliberate wrongdoing or gross negligence. They happen because the process had a gap — and nobody noticed until it was too late.
Understanding how failures occur is the first step toward preventing them. Here are the most common patterns.
The Most Common Root Causes
Data Entry Errors
When analysts enter results manually into spreadsheets or paper forms, transcription errors are inevitable at scale. A result of 0.05 mg/L entered as 0.5 mg/L changes the compliance picture entirely — and if no one is checking, it goes into the report, and from there into the regulatory record.
Hold Time Breaches
Many water quality parameters have strict hold times — maximum periods between sample collection and analysis. pH must be analyzed within 15 minutes of collection. Microbiological samples often have hold times of 6 to 24 hours. Missing these windows invalidates the result, but in a busy lab without automated tracking, samples can sit and nobody notices until after the analysis.
Missing Sign-offs
ISO 17025 and most national standards require that test results be reviewed and approved by a qualified second analyst before they are reported. When this happens over email or via a shared spreadsheet with no version control, sign-offs get missed, disputed, or simply undocumented.
Out-of-Spec QC Results Missed
Every batch of water tests should include quality control samples — blanks, duplicates, spikes, and certified reference materials. If a QC result is out of specification, the associated client results are compromised. But if QC data lives in a separate spreadsheet from sample data, the connection is easy to miss.
Report Version Confusion
Amended reports are a compliance risk that many labs underestimate. When a report is revised after initial issue — because of a corrected result, a client query, or a method revision — there must be a clear record of what changed and why. Without a LIMS, amended reports often overwrite originals, leaving no audit trail.
Failure Type Manual System Risk LIMS Prevention Data entry errors High — manual entry at every step Instrument integration removes manual transcription Hold time breaches High — no automated tracking Automated alerts before and at deadline Missing QC sign-offs Medium — email-based, undocumented Workflow blocks release without approval Out-of-spec QC unlinked High — separate files, no connection QC and sample data in one linked system Report version confusion High — overwrites common Full version history maintained automatically5. Core LIMS Features Every Water Lab Needs
Not every LIMS is built for the complexity of a water testing environment. Water testing involves a diverse range of parameter types — physical, chemical, microbiological, and radiological — each with their own testing methods, hold times, detection limits, and regulatory thresholds. A LIMS that works well for a clinical laboratory or a manufacturing QC lab may not serve a water lab's needs.
Here are the features that matter most.
Essential Feature Checklist
- Sample Registration with Barcode / QR Code Support — every sample must be uniquely identified from the moment it arrives, with collection details, client information, test parameters, and hold time clocks starting automatically
- Parameter-Specific Workflows — the system should support distinct workflows for drinking water, wastewater, process water, and environmental samples, each with its own approval sequences and reporting formats
- Instrument Integration — direct connection to laboratory instruments (ICP-MS, GC, HPLC, spectrophotometers, turbidimeters) eliminates manual transcription and reduces error
- QC sample management — built-in tracking for blanks, duplicates, matrix spikes, and certified reference materials, with automatic calculation of percent recovery and relative percent difference
- Hold Time Management — visual alerts and workflow blocks when samples are approaching or past their analytical hold times
- Chain of Custody Documentation — electronic COC forms that capture every transfer of sample custody, from collection through analysis and disposal
- Regulatory Report Templates — pre-built report formats for BIS, CPCB, EPA, EU Drinking Water Directive, and other applicable regulatory bodies
- Client Portal — secure, branded portal where clients can view and download their results in real time
- Multi-location Support — for labs operating from more than one site, centralized data management with location-specific access controls
- Audit Trail and Version Control — every action logged with user identification and timestamp; every report version stored permanently
When evaluating LIMS platforms for water testing, use this list as a non-negotiable baseline. A system that requires you to build hold time tracking or QC calculation logic from scratch is not ready for a water testing environment.
6. Sample Lifecycle Management
The sample is the fundamental unit of work in any testing laboratory. Everything — every workflow, every report, every compliance requirement — starts with a sample and ends with a verified result. A LIMS built for water testing tracks every stage of that journey.
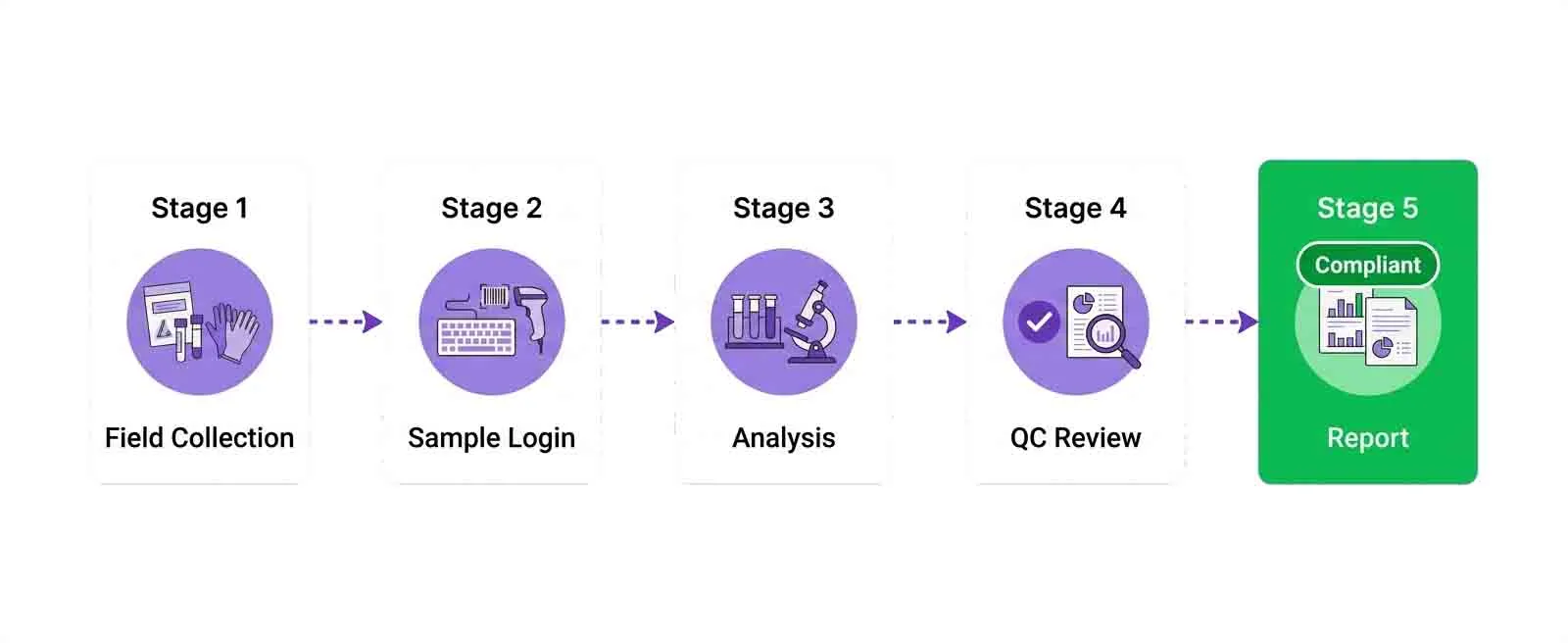
Stage 1 — Sample Collection and Registration
The lifecycle begins before the sample even arrives at the lab. Field technicians can register samples at the point of collection using a mobile interface, capturing GPS coordinates, collection time, sampler name, sample conditions (temperature, appearance), and the parameters to be tested. This information flows directly into the LIMS, triggering hold time clocks and creating the initial chain of custody record.
On arrival at the lab, samples are received, checked against the registration record, and assigned a unique LIMS identifier — typically a barcode or QR code. Any discrepancies between the field record and the physical sample are flagged immediately, before analysis begins.
Stage 2 — Test Assignment and Scheduling
Once registered, the LIMS automatically assigns samples to the appropriate test queues based on the requested parameters. A drinking water sample requiring pH, turbidity, total dissolved solids, coliform count, and heavy metals analysis will be routed to the relevant departments in the correct sequence, with scheduling optimized to meet the client's turnaround requirement.
Analysts see their work queue in real time. They can prioritize urgent samples, view hold time status, and access approved method documentation directly from the LIMS — no need to look up paper-based SOPs.
Stage 3 — Analysis and Data Capture
Results are entered into the LIMS either directly (for manual tests) or automatically via instrument integration. When data comes from an instrument, it is validated against preset acceptance criteria immediately. If a result falls outside the expected range — for example, a pH value that appears physically impossible — the system flags it for analyst review before it can progress.
Stage 4 — QC Review and Approval
Before results are released, they pass through a defined QC review stage. The second analyst or QC officer reviews the results alongside the associated QC data — blanks, duplicates, spikes. If everything is within specification, the approval is recorded digitally. If not, the batch is flagged for investigation and results are held until the issue is resolved.
Stage 5 — Report Generation and Delivery
Once approved, the LIMS generates the final report automatically, applying the correct template for the client and the regulatory framework. Reports include all required metadata — sample ID, collection details, analytical method, analyst name, QC data, detection limits, and regulatory compliance status for each parameter.
Reports are distributed electronically, delivered to the client portal, and stored permanently in the LIMS with a version record. The entire lifecycle — from field collection to report delivery — is documented and traceable.
7. Quality Control — Built Into the Workflow
This is one of the most significant differences between a manual system and a well-configured LIMS.
QC Sample Types in Water Testing
- Method Blanks — samples of pure water processed through the entire analytical method to detect contamination from reagents, vessels, or the environment,/li>
- Field Blanks — blank samples collected in the field, used to detect contamination introduced during sample collection or transport
- Duplicate Samples — two samples collected from the same source at the same time, analyzed separately, to assess measurement precision
- Matrix Spikes — samples spiked with a known concentration of the target analyte, used to assess the effect of the sample matrix on recovery
- Certified Reference Materials — samples with certified concentrations, analyzed regularly to verify instrument calibration and method performance
How LIMS Automates QC
In a LIMS, QC samples are not optional additions — they are required components of every analytical batch. The system will not allow a batch to proceed to reporting unless the required QC samples are included and their results are within specification.
- QC samples are registered and tracked through the same sample lifecycle as client samples
- Results are automatically compared against pre-set acceptance criteria
- Out-of-spec QC results trigger an alert and place the associated client results on hold
- The QC investigation is documented within the LIMS before results can be released
- All QC data is permanently linked to the client results and included in the audit trail
Why This Matters for Accreditation
ISO 17025 assessors specifically look for evidence that QC data is linked to analytical results and that out-of-spec QC results trigger documented investigations. A LIMS provides this evidence automatically — without the lab having to reconstruct it from paper records at assessment time.
8. Regulatory Reporting Made Simple
Regulatory reporting is where compliance becomes visible. It is the moment when the lab's work — every sample received, every test performed, every result verified — is translated into a formal document that is submitted to a client, a regulatory authority, or an accreditation body.
Getting this right is non-negotiable. A report with incorrect formatting, missing data elements, or inconsistent result presentation can be rejected — or worse, queried after submission, triggering an audit.
What Regulators Actually Require
While specific requirements vary by jurisdiction, most regulatory frameworks for water quality reporting require the following elements in every formal report:
- Laboratory identification and accreditation number
- Client information and sample source identification
- Date and time of sample collection, receipt, and analysis
- Analytical method reference (e.g., IS, EPA, APHA, ISO method number)
- Result for each parameter, with unit of measurement
- Method detection limit and reporting limit
- Regulatory limit for each parameter (where applicable)
- Compliance status — compliant or non-compliant — for each parameter
- Statement of uncertainty (for ISO 17025 accredited labs),/li>
- Analyst signature and reviewer approval
- Report date and unique report identifier
How LIMS Simplifies Reporting
A LIMS with regulatory reporting capability stores all of the above information as structured data, linked to the sample and the analytical result. Report generation is not a manual task — it is an automated output that pulls the correct data elements into the correct template, applies the correct formatting, and checks for completeness before the report can be finalized.
This means that generating a fully compliant regulatory report takes minutes, not hours. It also means that reports are consistent — every report generated for a given regulatory authority looks the same, contains the same elements, and meets the same standard. There is no risk of a format error from a tired analyst working on their fifteenth report of the day.
Amended Reports and Version Control
When a result needs to be amended after a report has been issued — a situation that arises in every laboratory eventually — the LIMS creates a new report version automatically. The original report is preserved. The amendment is documented, with a reason code, the identity of the person who approved it, and the date. Both versions are permanently stored and can be retrieved instantly if the change is ever questioned.
9. Chain of Custody — The Audit Trail That Protects You
Chain of custody is the documented record of who has had control of a sample, when, and under what conditions, from the moment of collection to the moment of disposal. In water testing, it serves two purposes: it demonstrates the integrity of the sample, and it protects the laboratory from disputes about results.
In a legal or regulatory dispute — for example, an industrial client challenging a non-compliant discharge result — the chain of custody is often the most important piece of evidence the lab can produce. If it is incomplete, or if it exists only as a handwritten form that has been scanned and filed somewhere on a shared drive, it may not hold up to scrutiny.
What a Digital COC Captures
- Sample collector name, ID, and contact information
- Collection location (GPS coordinates where applicable)
- Date, time, and conditions at collection
- Container type, preservative used, and seal integrity confirmation
- Transport conditions (temperature, courier details)
- Receiving lab technician name and time of receipt
- Storage location and conditions within the lab
- Every analyst who touched the sample, and when
- Transfer records if the sample was sent to a sub-contract lab
- Disposal date, method, and authorization
In a LIMS, this information is captured at each transition — not reconstructed afterward. When a sample moves from receiving to storage, the LIMS records it. When an analyst removes a sample from storage for testing, the LIMS records it. When the sample is disposed of after the retention period, the LIMS records it. The result is a continuous, unbroken audit trail that requires no manual maintenance.
Practical Example
A water testing lab receives a complaint from a municipal client claiming that a reported lead result of 0.018 mg/L (above the 0.010 mg/L regulatory limit) was caused by contamination during sample handling. With a full LIMS-generated chain of custody, the lab can demonstrate exactly who collected the sample, what containers were used, what the transport conditions were, how quickly the sample was analyzed, and which QC samples were run alongside it — turning a potential dispute into a documented defense.
10. LIMS vs. Manual Systems — A Practical Comparison
For laboratory managers who are evaluating the case for LIMS investment, a direct comparison between the operational realities of manual and LIMS-driven systems is the most useful starting point. The following scenarios illustrate the difference in practical terms.
Scenario A — High-Volume Day
The lab receives 150 samples on a Monday morning. In a manual system: reception logs each sample in a spreadsheet, prints work orders, and distributes them by department. Analysts pull samples from the fridge, check the paper work order, complete their analysis, and enter results into a department spreadsheet. A senior analyst consolidates all results into a master file. The QC officer checks the QC samples and initials a printed form. The report writer copies results into a Word template and emails the finished report.
In a LIMS: samples are scanned on arrival, hold time clocks start automatically, work queues populate in each department, instrument results transfer directly to the system, QC is checked against pre-set criteria in real time, and reports are generated with one click from the LIMS dashboard. The entire process is faster, more accurate, and leaves a complete audit trail.
Scenario B — Pre-Audit Preparation
A regulatory inspector has announced a site visit. In a manual system: the QA manager spends several days gathering COC forms, QC logs, analyst sign-off records, and report copies from various filing systems. Some documents are missing. Some QC forms are incomplete. The audit is stressful and results in several corrective action requests.
In a LIMS: the QA manager runs a pre-configured audit report in minutes. Every sample record, QC result, approval, and report version is available in the system, searchable and filterable by date, parameter, analyst, or client. The audit preparation takes an afternoon instead of a week.
| Activity | Manual System | LIMS |
|---|---|---|
| Sample registration (150 samples) | 2–3 hours | 30–45 minutes |
| QC check on a batch | Manual calculation, 45 min | Automatic, real-time |
| Report generation | 1–2 hours per report | 5–10 minutes per report |
| Audit preparation | 3–5 days | 2–4 hours |
| Amended report | Manual re-creation, version risk | New version auto-created, original preserved |
11. Choosing the Right LIMS for Your Water Lab
The LIMS market has expanded significantly, and not every platform is designed for water testing. Some are built for clinical environments, others for pharmaceutical manufacturing, and others for general chemistry. A water testing lab needs a system that understands its specific workflows, parameters, and regulatory context.
Here is a structured approach to evaluating LIMS options.
Step 1 — Define Your Requirements
Before looking at software, document your lab's current state and future needs:
- What types of samples do you receive? (Drinking water, wastewater, industrial effluent, environmental)
- How many samples do you process per day, per week, per month?
- What regulatory frameworks do you report to? (BIS, CPCB, EPA, EU DWD, ISO 17025, NABL)
- How many analysts and departments do you have?
- Do you operate from a single site or multiple locations?
- What instruments do you need to integrate?
- Do your clients require a portal for real-time access to results?
Step 2 — Evaluate Core Capabilities
With your requirements documented, evaluate LIMS platforms against these non-negotiable capabilities for water testing:
- Water-specific parameter libraries — the system should include standard parameter lists for drinking water, wastewater, and environmental monitoring without requiring you to build them from scratch
- Hold time management — automated tracking and alerting, not a manual calendar
- Instrument integration — direct interface with your existing instruments
- QC workflow enforcement — the system must prevent reporting without completed QC
- Regulatory report templates — pre-built, not custom-coded by your IT team
- Audit trail — comprehensive, automatic, and accessible
Step 3 — Assess Vendor Support and Implementation
A LIMS implementation is a significant operational project. The quality of the vendor's implementation support often matters more than the software features. Ask vendors:
- What does the implementation timeline look like for a lab our size?
- How is training delivered, and over what period?
- What ongoing support is available after go-live?
- Can you provide references from water testing labs of similar scale?
12. Implementation — What the Journey Looks Like
One of the most common reasons labs delay LIMS adoption is concern about disruption. The fear is understandable — the lab cannot stop operating while a new system is installed. Samples arrive every day. Reports are due. Clients cannot wait.
A well-managed LIMS implementation addresses this directly. The goal is not to switch everything over on day one — it is to build the new system alongside the existing process, transfer knowledge and data in stages, and cut over when confidence is high.
Typical Implementation Phases
Phase 1 — Discovery and Configuration (Weeks 1–4)
The implementation team works with the lab's QA manager and department heads to document existing workflows, test menus, method references, regulatory requirements, and client reporting formats. This information is used to configure the LIMS before any live data entry begins. At the end of this phase, the lab has a configured system that mirrors its workflows — not a generic template.
Phase 2 — Validation and Testing (Weeks 5–8)
Before the LIMS goes live, it is tested against real-world scenarios from the lab's operations. Analysts run test samples through the system, generate mock reports, simulate out-of-spec QC situations, and check that hold time alerts work correctly. Any configuration adjustments are made at this stage.
Phase 3 — Training (Weeks 7–10)
Training is delivered in parallel with the final testing phase, department by department. Reception staff learn sample registration. Analysts learn result entry and QC workflows. QA officers learn the review and approval process. Lab managers learn the reporting and administration functions. Training is hands-on, using the lab's own data and workflows.
Phase 4 — Parallel Running (Weeks 9–12)
For two to four weeks, the lab runs both the existing manual system and the LIMS simultaneously. Every sample is processed in both systems, and results are compared. This phase builds confidence, identifies any remaining configuration gaps, and ensures that the team is comfortable before full transition.
Phase 5 — Go-Live and Stabilization (Week 12 onwards)
The manual system is retired. The LIMS becomes the system of record. Vendor support is available during the stabilization period — typically 30 to 60 days — to address any issues that arise in live conditions.
Implementation Reality Check
Most water testing labs complete LIMS implementation without significant disruption to operations. The parallel running phase is the key — it removes the risk from the cutover and gives the team genuine confidence in the new system before the old one is switched off.
13. ROI — The Business Case for LIMS
For laboratory directors and business owners evaluating LIMS investment, the question is not just whether the system improves compliance — it is whether it delivers measurable value. The answer, across virtually every water testing lab that has made the transition, is yes.
The return on investment comes from multiple directions.
Direct Cost Savings
Time Savings on Reporting
In a water testing lab processing high sample volumes, manual report generation — pulling results from multiple spreadsheets, populating a Word template, formatting for regulatory submission — routinely takes an hour or more per client report. With a LIMS, the same output is generated in 10–15 minutes. For a lab issuing a significant number of reports daily, the cumulative time reclaimed is substantial.
Reduction in Rework
Compliance failures, amended reports, and QC failures that require reanalysis are expensive. They consume analyst time, use reagents and standards, and delay delivery to clients. Labs that transition from manual systems to LIMS consistently report a meaningful reduction in rework rates within the first six months of operation — driven primarily by instrument integration eliminating transcription errors and automated QC checks catching out-of-spec results before they reach the report stage.
Audit Preparation Costs
Preparing documentation for an ISO 17025 surveillance audit or a regulatory inspection can take a QA team several working days. With LIMS, this time is reduced to hours. For labs that undergo multiple audits per year, this is a meaningful cost reduction.
Revenue Protection and Growth
Beyond cost savings, LIMS protects and enables revenue in ways that are harder to quantify but equally significant.
- Accreditation protection — maintaining ISO 17025 or NABL accreditation is a prerequisite for many contracts. A compliance failure that results in suspension of accreditation can cost a lab its largest clients.
- Faster turnaround — with LIMS, labs can credibly offer faster turnaround times without increasing error risk. This is a competitive advantage in a market where clients increasingly expect next-day results.Capacity for growth — a manual system that is already at its limits cannot easily scale. A LIMS scales with the lab — adding analysts, instruments, and sample types without proportional increases in administrative overhead.
- Assess your current system — document the pain points your team experiences today around sample tracking, QC management, reporting, and audit preparation. These pain points define the value LIMS will deliver for your specific operation.
- Define your compliance requirements — list the regulatory frameworks and accreditation standards that apply to your lab. These define the non-negotiable features your LIMS must support.
- Evaluate your options — request demonstrations from LIMS vendors who have specific experience with water testing laboratories. Ask to see water-specific workflows, not generic demos.
- Build your internal business case — use the ROI framework from Section 13 to estimate the time and cost savings that LIMS would deliver for your lab's volume and workflow.
- Plan your implementation — work with your chosen vendor to build a realistic implementation plan that minimizes disruption to ongoing operations.
| Value Driver | Manual System | With LIMS |
|---|---|---|
| Report generation time (per report) | 45–90+ min | 10–15 min |
| Rework rate (QC/data errors) | Elevated — manual entry at every step | Significantly lower — instrument integration and QC enforcement remove primary error sources |
| Audit prep time | 3–5 days | Half a day |
| Sample turnaround capacity (daily) | Limited by admin | Scales with instruments |
14. What a Purpose-Built Water Lab LIMS Looks Like in Practice
The previous sections have outlined what a water testing lab needs from a LIMS. This section describes what it looks like when a platform actually delivers on those requirements — not as a feature list, but as a practical account of how the right system behaves in a real laboratory environment. Use it as a reference when evaluating vendors and configuring demonstrations.
Pre-configured for water testing, not adapted from somewhere else
A LIMS purpose-built for water testing should arrive with parameter libraries already populated for drinking water, wastewater, industrial effluent, and environmental monitoring. You should not be building your test menu from a blank list, or asking your IT team to configure hold time alerts from scratch. The system should understand the language of water testing from day one — parameter names, units, regulatory thresholds, method references — because these are structural to the software, not an add-on layer.
Generic LIMS platforms — those designed primarily for pharmaceutical manufacturing, clinical environments, or broad QC applications — often require extensive customisation before they can handle the specific workflows of a water testing lab. That customisation is expensive, time-consuming, and frequently incomplete. The right water lab LIMS should not require you to describe what a matrix spike is.
QC that enforces itself
Percent recovery and relative percent difference (RPD) calculations should be automated. When a matrix spike or field duplicate falls outside acceptance criteria, the system should immediately place the associated client results on hold — without anyone having to remember to check. QC is not a module you activate; it is the way the system processes every batch.
Regulatory reports that go out in minutes
Report templates for the frameworks your lab works under — BIS, CPCB, EPA, ISO 17025, NABL, EU Drinking Water Directive — should be ready to use on day one, not pending customisation. The system should populate every required data element automatically, flag any missing fields before the report is finalised, and maintain a permanent version record every time an amendment is made. Generating a fully compliant regulatory report should take minutes, and the output should be identical in structure and content every time.
An audit trail that requires no maintenance
Every analyst action, every approval, every result amendment, every chain of custody transfer should be recorded automatically with a timestamp and user identification — without anyone having to log it manually. When a regulatory inspector asks for records from a specific date range, the answer should be a filtered search that takes seconds. The value of a good audit trail is that you never have to think about it until you need it — and then it is exactly there.calability without administrative overhead
Whether a lab is processing 30 samples a day or 3,000, the core workflow should function the same way. A LIMS built for growth should handle multi-site operations with centralised data management and location-specific access controls, support instrument integration as the lab’s instrument portfolio expands, and scale sample throughput without proportional increases in administrative staff. The system should grow with the lab — not become a bottleneck that forces the lab to grow around it.
The platforms that deliver on all of the above are the ones worth evaluating seriously. A structured demonstration — configured for your lab’s own parameters, methods, and regulatory requirements — is the most reliable way to determine whether a system genuinely meets your operational needs or merely claims to. www.revolims.com offers exactly this kind of workflow-specific demonstration for water and environmental testing laboratories.
15. Conclusion & Next Steps
The compliance challenge facing water testing labs in 2026 is real. Regulations are getting stricter, sample volumes are increasing, and clients and regulators alike expect faster, more transparent reporting. The labs that are navigating this environment successfully have one thing in common: they have built operational systems that make compliance a natural outcome of daily work — not an afterthought.
A purpose-built LIMS is the most direct path to that outcome. It transforms sample management from a manual, error-prone process into a structured, traceable workflow. It builds QC into every analytical batch. It generates regulatory reports automatically. It maintains the audit trail without anyone having to maintain it. And it scales — so the lab can grow its capacity without proportionally growing its administrative burden.
The question for laboratory leaders is not whether LIMS delivers value. The evidence on that is clear. The question is where the lab is in its readiness to make the transition — and what the first practical step looks like.
Your Next Steps
Frequently Asked Questions
What is a LIMS and why do water testing labs need one?
A Laboratory Information Management System (LIMS) is software that manages the complete lifecycle of samples and laboratory data — from registration and test assignment through analysis, QC review, reporting, and audit trail maintenance. Water testing labs need a LIMS because manual systems based on spreadsheets and paper cannot reliably enforce hold times, link QC data to client results, or generate the structured digital reports now required by regulators and accreditation bodies. As sample volumes and regulatory complexity increase, a LIMS becomes the operational backbone that makes compliance a consistent, repeatable outcome.
How does LIMS support ISO 17025 compliance for water labs?
ISO 17025 requires documented procedures, full measurement traceability, regular equipment calibration records, defined QC protocols, and comprehensive record-keeping for every test. A LIMS addresses all of these requirements structurally — audit trails are automatic and tamper-evident, QC data is linked directly to client results, calibration records are stored in the system, and every analyst action is logged with a timestamp. During accreditation assessments, labs running on LIMS can retrieve any record instantly rather than reconstructing it from paper files, which significantly reduces the risk of corrective action findings.
What is hold time management in a water testing LIMS?
Hold time management refers to the automated tracking of the maximum permitted period between sample collection and analysis for each parameter. Different parameters have different hold times — pH must typically be analysed within 15 minutes of collection, while many chemical parameters allow up to 28 days. In a manual system, hold time tracking is a manual calendar task prone to error in busy laboratory environments. A LIMS starts the clock automatically at sample registration and sends visual alerts as deadlines approach, preventing the analysis of invalidated samples and protecting the integrity of results submitted to clients and regulators.
What is chain of custody and how does LIMS manage it?
Chain of custody (COC) is the documented record of everyone who has had control of a sample — from the moment of collection through laboratory analysis to final disposal. In water testing, it is a regulatory requirement and a critical defence in any dispute about results. A LIMS captures COC data at each transition automatically: field collection, receipt at the lab, movement between departments, sub-contract transfers, and sample disposal. This creates a continuous, unbroken record that can be produced instantly during an audit or legal challenge, without relying on handwritten forms or scanned documents.
How long does LIMS implementation take for a water testing lab?
A structured LIMS implementation for a water testing lab typically takes 10 to 14 weeks from initial discovery to go-live, depending on the lab’s size, complexity, and the number of instrument integrations required. The process is divided into discovery and configuration, validation and testing, training, parallel running (where both the old and new systems operate simultaneously to build confidence), and stabilisation after cutover. Most labs complete the transition without significant disruption to ongoing operations, largely because a well-managed parallel running phase removes the risk from the final switchover.
Can a LIMS generate reports for BIS, CPCB, EPA, and other regulatory bodies?
Yes — a purpose-built water testing LIMS should include pre-configured report templates for the major regulatory frameworks applicable to water quality testing, including BIS (IS 10500) for Indian drinking water standards, CPCB guidelines for environmental discharge, EPA methods under the Safe Drinking Water Act, and ISO 17025 report formats. These templates pull all required data elements automatically — accreditation numbers, method references, detection limits, compliance status per parameter, analyst sign-off — and generate a fully formatted, submission-ready document without manual assembly.
Water testing laboratories exploring LIMS adoption can request a workflow-specific demonstration at www.revolims.com — configured for your parameters, your regulatory frameworks, and your reporting requirements.
Author: Revol LIMS Team





















