
The GCC Petrochemical Lab's Digital Moment — And How to Make the Most of It
The GCC's petrochemical sector is in the middle of one of the most ambitious industrial transformations in its history. Facilities across Jubail, Ruwais, Sohar, and beyond are expanding capacity, diversifying product portfolios, and integrating into global supply chains that demand higher standards of quality documentation, traceability, and regulatory alignment than ever before.
In this environment, the laboratory sits at a critical junction. The QC lab is where raw materials are qualified, in-process parameters are confirmed, and finished products are released. It is, in a very real sense, the gatekeeping function that everything downstream depends on.
The facilities that recognize this — and invest in making their lab data infrastructure match the sophistication of their broader operations — are positioning themselves for significant advantage: faster regulatory clearances, stronger audit outcomes, better integration with digital supply chain partners, and more agile responses to the quality demands of international customers.
This article is about what that investment looks like in practice, why the timing is right, and where a Laboratory Information Management System fits into the broader digitalization picture.
The GCC Lab's Evolving Regulatory Context
Understanding the regulatory environment is the starting point for any meaningful conversation about lab data infrastructure in the GCC.
The Saudi Food and Drug Authority has matured significantly as a regulatory body over the past several years. Its achievement of WHO Maturity Level 4 recognition — a designation held by a small number of regulatory authorities globally — reflects an inspection capability and framework sophistication that is now comparable to leading international agencies. For labs operating under SFDA oversight, this translates to a growing expectation of data integrity, electronic record-keeping, and traceable audit trails.
ISO/IEC 17025 accreditation has become the standard baseline for industrial QC labs across the region. The UAE's Emirates International Accreditation Centre, Saudi Arabia's Saudi Accreditation Centre, and equivalent bodies in Qatar, Oman, Kuwait, and Bahrain all operate frameworks in which data integrity and measurement traceability are foundational requirements — not optional enhancements.
What this means practically is that GCC labs are being evaluated not just on the quality of their results, but on the quality of the data chain that produced them. Traceability from sample receipt through analysis to final report, instrument calibration records linked to every result, and electronic audit trails that can be queried during an inspection are increasingly the expected standard.
This is good news for facilities that are ready for it. A lab that can demonstrate this level of data governance is not just audit-ready — it is positioned as a credible quality partner for international customers, multinational joint ventures, and supply chain relationships where data integrity documentation is a qualification requirement.
Where the Opportunity Is
Across the GCC, the conversation about digital transformation in industrial operations has been running at the strategic level for several years. Saudi Vision 2030's industrial diversification agenda, the UAE's Operation 300bn manufacturing strategy, and Qatar's National Vision 2030 are all underpinned by a clear expectation of digital-first operations.
What is increasingly clear is that this transformation is moving from strategy to execution. Majors like Saudi Aramco, ADNOC, and SABIC have active digital transformation programs that are reshaping how they manage operational data, and the same expectations are flowing through to their supply chains and joint venture partners. When a major downstream customer asks for digital quality records, traceable test results, and electronic certificates of analysis — and that request is coming more frequently — a paper-based lab record system creates friction that a digital one doesn't.
The opportunity here is not simply to become compliant. It is to become the kind of quality partner that the GCC's most demanding industrial relationships require.
Lab digitalization, done well, delivers three distinct benefits that compound over time:
Regulatory confidence. When the next ISO 17025 surveillance audit, SFDA inspection, or customer quality audit arrives, a digitally managed lab responds with data that is immediately queryable, complete, and tamper-evident. Preparation time compresses dramatically. Findings related to record-keeping and traceability — among the most common sources of observations across accredited labs — become significantly less likely.
Operational efficiency. The time that analysts spend on manual transcription, paper record maintenance, and report compilation is real. In a region where developing and retaining qualified lab talent is a genuine operational priority, redirecting that time toward analysis, method development, and quality oversight is a meaningful gain.
Integration readiness. The broader digital transformation agenda — IoT sensor networks, real-time operational dashboards, ERP integration, predictive quality analytics — all depends on a foundational layer of clean, structured, digital lab data. A Laboratory Information Management System is that foundation. Without it, the more advanced integrations either don't work or require costly workarounds.
Understanding the Lab Data Gap
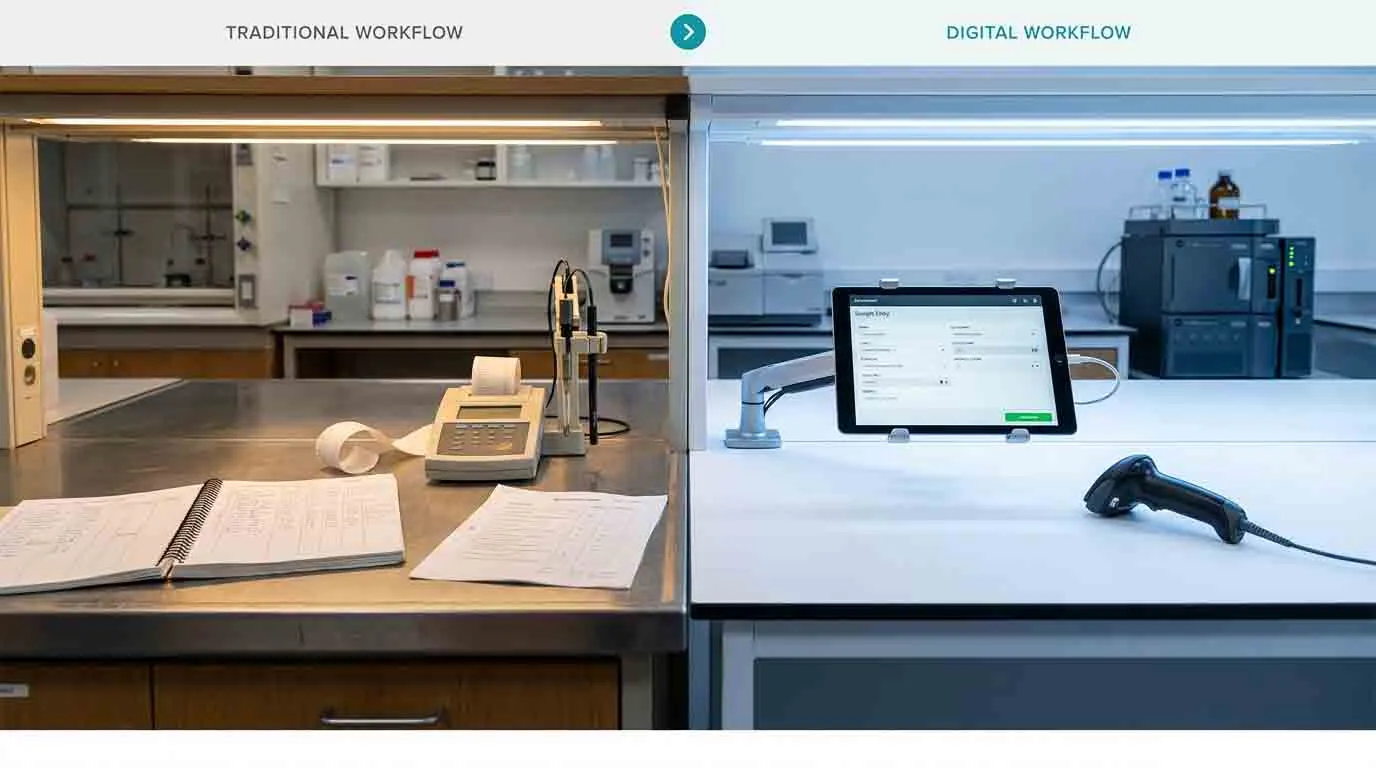
For many GCC industrial labs, there's a gap between the sophistication of their analytical capabilities and the sophistication of their data management infrastructure. The instruments may be current. The methods may be validated. The analysts may be skilled. But somewhere in the workflow, data is still moving through manual steps — handwritten sample logs, transcription from instrument printouts, paper batch records, spreadsheet-based result tracking.
The most common points in a petrochemical QC lab workflow where manual data handling creates avoidable exposure are:
Sample login and chain of custody. When samples arrive and are logged manually, the record chain depends entirely on the accuracy and consistency of individual data entry. In high-volume labs, this is where errors accumulate most quickly, and where an auditor is most likely to find gaps.
Result transcription. When an analyst records an instrument result on paper and then transfers it to a spreadsheet or logbook, every transcription step is a point where a digit can be transposed, a unit misread, or a value accidentally overwritten. More importantly, the transcribed result loses its connection to the instrument that generated it — which is precisely the traceability link that ISO 17025 and SFDA data integrity expectations require.
Out-of-specification management. When a result falls outside specification, the response workflow — documentation, investigation, disposition — needs to be traceable and complete. In a paper-based system, this depends on individual discipline and institutional memory. In a digital system, it's triggered automatically and tracked to closure.
Calibration and instrument qualification records. Maintaining the link between a test result and the calibration status of the instrument that produced it is a core ISO 17025 requirement. Doing this manually, across a fleet of instruments on different calibration schedules, is administratively intensive and prone to gaps under operational pressure.
None of these gaps are unique to GCC facilities — they're common across industrial labs globally. What makes them particularly relevant in the current GCC context is the tightening regulatory environment and the increasing quality expectations of international commercial relationships.
What a LIMS Does — and Why It's the Right Starting Point
A Laboratory Information Management System is, at its core, the system of record for everything that happens inside a lab. It manages the full lifecycle of a sample — from registration and assignment through analysis, result validation, and final reporting — with a complete electronic record at every step.
For a GCC petrochemical QC lab, the practical impact looks like this:
When a sample arrives, it is registered in the LIMS with a unique identifier, linked to its source and the tests required. When it is assigned to an analyst, that assignment is recorded. When the analysis is complete, the result is entered directly — or captured automatically from the instrument — and compared against specification limits in real time. If it passes, it moves through an electronic review and approval workflow. If it doesn't, an OOS investigation is triggered automatically. The final certificate of analysis is generated from the validated data, not typed from scratch.
At every step, the record is timestamped, linked to the relevant analyst qualifications and instrument calibration status, and stored in a system that maintains a complete, uneditable audit trail.
For ISO 17025 and SFDA compliance purposes, this is the architecture that makes audit preparation a reporting exercise rather than a reconstruction effort. The data that an auditor needs is already organized, queryable, and complete.
For operational purposes, it means that quality decisions can be made on current data, not data that is hours old. Out-of-specification results are visible immediately. Trends across batches and shifts are accessible in real time. The lab manager has a live view of lab status without waiting for paper records to be compiled.
For integration purposes, it means that the rest of the digital infrastructure — IoT platforms, ERP systems, supply chain portals — has a reliable, structured data source to connect to.
Selecting and Deploying a LIMS in the GCC Context
The right LIMS for a GCC petrochemical lab isn't necessarily the most complex one. It's the one that fits the lab's actual workflow, meets the applicable regulatory requirements, and can be deployed and validated within a realistic timeframe and budget.
A few considerations that are particularly relevant in the GCC context:
Deployment model and data residency. Saudi Arabia's National Data Governance Framework and the UAE's data residency requirements mean that public cloud-only LIMS deployments may not be appropriate for all GCC facilities. A LIMS that offers on-premise or private cloud hosting options — without sacrificing functionality — is often the right fit for regulated industrial environments in this region. This is a question worth asking directly at the evaluation stage.
Validation documentation. For facilities operating under SFDA oversight or ISO 17025 accreditation, the LIMS itself needs to be validated as a system that produces reliable, accurate results. A reputable LIMS provider will have IQ/OQ/PQ documentation frameworks, risk assessments, and validation support built into the implementation process — not offered as an optional extra.
Configuration depth for petrochemical workflows. Industrial QC labs in the petrochemical sector have specific requirements: multi-parameter sample types, complex specification structures, shift-based reporting, integration with process historians and ERP systems. The LIMS should accommodate these through standard configuration, not bespoke development. A system that requires significant custom coding to meet standard QC requirements will create long-term maintenance challenges.
Local and regional implementation support. Deployment quality depends significantly on the implementation team's familiarity with the regulatory context and operational environment. For GCC facilities, working with a LIMS provider that has regional experience — and can provide on-the-ground support rather than remote-only implementation — reduces deployment risk meaningfully.
Phased implementation. The most successful LIMS deployments in industrial labs are phased rather than big-bang. Starting with the highest-value workflows — typically sample management and result entry — allows the lab to build confidence and demonstrate value before expanding scope. A provider that encourages a phased approach is usually more interested in a successful outcome than a large upfront contract.
Frequently Asked Questions
How long does a LIMS implementation typically take for an industrial lab?
For a QC lab with defined scope and a phased approach, most facilities reach operational stability within three to six months of go-live. Larger, more complex deployments with multiple sites or extensive instrument integrations may take longer — but the key is that value is being delivered from the first phase, not at the end of a long monolithic project.
Does a LIMS replace existing ERP or process control systems?
No — a LIMS complements them. The LIMS manages lab-specific data: samples, results, methods, instruments, and analysts. ERP systems manage production planning, inventory, and commercial transactions. Process control systems manage real-time plant operations. A LIMS connects to both, providing the quality data layer that neither was designed to handle natively.
What does the transition look like for lab staff?
For most analysts, the transition from a paper-based system to a well-configured LIMS is straightforward — the system guides them through the workflow they already follow, but digitally. The more significant change is for lab supervisors and managers, who gain real-time visibility they didn't have before. Most labs find that adoption improves quickly once staff experience the reduction in administrative overhead.
How does a LIMS support multi-site operations?
A centralized LIMS provides a single data environment across multiple lab locations — consistent methods, unified reporting, and organization-wide compliance visibility. For GCC operators with facilities in Saudi Arabia, the UAE, and elsewhere in the region, this is one of the more significant operational benefits.
The Competitive Position That Digital Lab Data Creates
The GCC petrochemical sector is competing on the global stage. The facilities that win long-term supply chain partnerships with international customers, that clear regulatory reviews efficiently, and that scale quality operations across multiple sites without proportional overhead growth are the ones that have invested in the right infrastructure at the right time.
Lab data management is part of that infrastructure. A QC lab that delivers traceable, audit-ready, digitally managed quality records isn't just operationally efficient — it is demonstrably positioned as a serious quality partner. That positioning has commercial value that extends well beyond the lab itself.
The digital transformation of the GCC's industrial sector is already underway. For labs that have not yet made the transition to digital data management, the question is simply where in that journey to begin — and which starting point delivers the clearest early value.
For most QC labs in the petrochemical sector, the answer is sample management, result traceability, and OOS workflow. Get those right, digitally, and the rest of the journey follows logically.
Ready to explore what a LIMS deployment looks like for your GCC facility?
[Book a 20-minute discovery call with our team].
Author: Revol LIMS Team





















