
The Future is Agentic: How Autonomous LIMS Systems Will Transform Laboratory Workflows
A new class of artificial intelligence — one that perceives, plans, decides, and acts — is making its way into the laboratory. Here is what it means for your operations, and why Revol LIMS is already building it.
TABLE OF CONTENTS
▸ 2. What Agentic AI Actually Means — and What It Does Not
▸ 3. The Four Pillars of an Agentic LIMS
▸ 4. How Autonomous LIMS Transforms Laboratory Workflows
▸ 5. Agentic AI, Compliance, and the Question of Trust
▸ 6. Where Revol LIMS Stands Today
▸ 7. How to Prepare Your Lab for the Agentic Era
▸ 8. Conclusion
For most of its history, the Laboratory Information Management System lived comfortably in the role of a highly organized record-keeper. It tracked samples, logged results, enforced workflows, and ensured that nothing fell through the cracks of a busy lab. That was — and still is — genuinely valuable. But it was, at its core, reactive. A LIMS did what it was told, when it was told.
A profound shift is now underway. The same wave of artificial intelligence that gave us generative text, predictive analytics, and natural language interfaces is evolving into something qualitatively different: agentic AI. This is AI that does not simply respond to prompts. It perceives its environment, understands goals, forms plans, executes multi-step actions, monitors outcomes, and adjusts — autonomously — until the goal is achieved.
For laboratory operations, the implications are enormous. An agentic LIMS would not just remind a technician that a sample is overdue; it would investigate why, reschedule the relevant test, notify the appropriate personnel, update the client portal, and log every decision it made — all without a single human command. That is the laboratory of the near future. And at Revol LIMS, we are already building it.
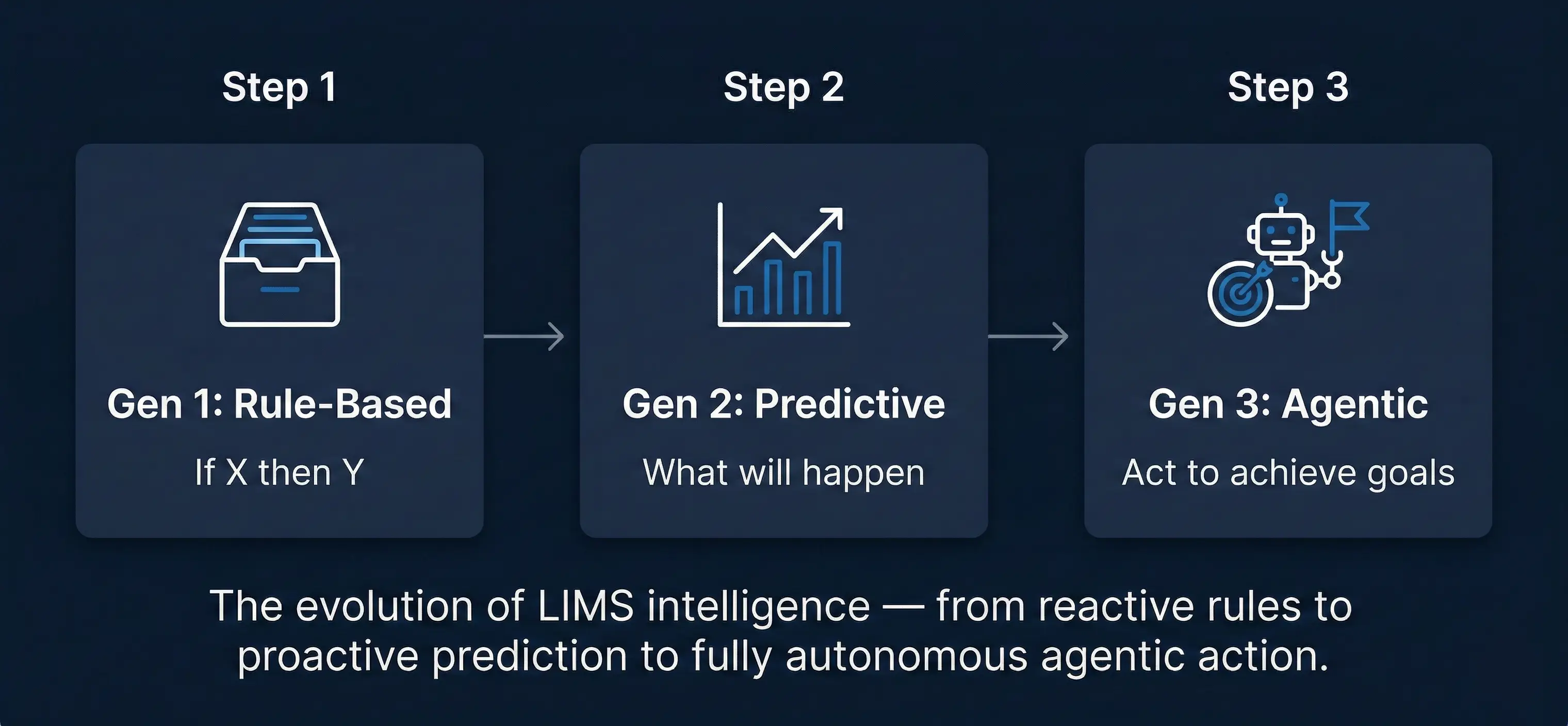
1. From Reactive to Agentic: A Fundamental Shift in Lab AI
To understand what agentic AI represents, it helps to trace the generational arc of intelligence in LIMS platforms.
The first generation of 'smart' LIMS added rule-based automation — if a result exceeded a control limit, send an alert. Useful, but brittle. Rules had to be written manually, and they could not anticipate scenarios their authors had not imagined.
The second generation brought machine learning and predictive analytics. Rather than following static rules, the system could learn patterns from historical data and forecast outcomes. A LIMS could now say: Based on current trends, this instrument will likely require recalibration within three days. This was a genuine leap forward, enabling laboratories to shift from reactive problem-solving to proactive quality management.
The third generation — and the one that defines the next decade — is agentic. Here, the system is not just predicting; it is deciding and doing. It understands the lab's objectives (maximize throughput, maintain compliance, minimize rework) and actively pursues them, coordinating actions across workflows, instruments, personnel, and external systems.
"The difference between predictive AI and agentic AI is the difference between a weather forecast and a self-driving car. One tells you what is coming. The other navigates you through it."
2. What Agentic AI Actually Means — and What It Does Not
The term 'agentic AI' has entered mainstream technology discourse rapidly enough that some precision is warranted. An agentic AI system is characterized by four defining properties: goal-directedness, perception, planning, and autonomous action.
Goal-directedness means the system has an objective it works toward over time — not just a prompt it responds to once. In a laboratory context, the goal might be to ensure all priority samples are reported within a defined turnaround time, or to maintain a Six Sigma quality level across a specific product line.
Perception means the system continuously reads its environment — sample queues, instrument statuses, personnel schedules, QC trends, client deadlines, regulatory calendars — and interprets what it observes in the context of its goals.
Planning means the system generates a sequence of actions to achieve its goal, considers alternatives, weighs constraints, and selects the most effective path forward. Unlike a simple automation script, a planning agent can handle novel situations that no rule was explicitly written for.
Autonomous action means the system can execute those plans — logging data, triggering workflows, communicating with instruments, generating reports, notifying stakeholders — without waiting for a human to click a button at every step.
What agentic AI is not is unbounded or unaccountable. Every action is logged, every decision is auditable, and human oversight is built into the architecture at configurable checkpoints. In a regulated laboratory environment, this is not optional — it is foundational. Agentic systems in LIMS are designed to amplify human judgment, not replace it.
3. The Four Pillars of an Agentic LIMS
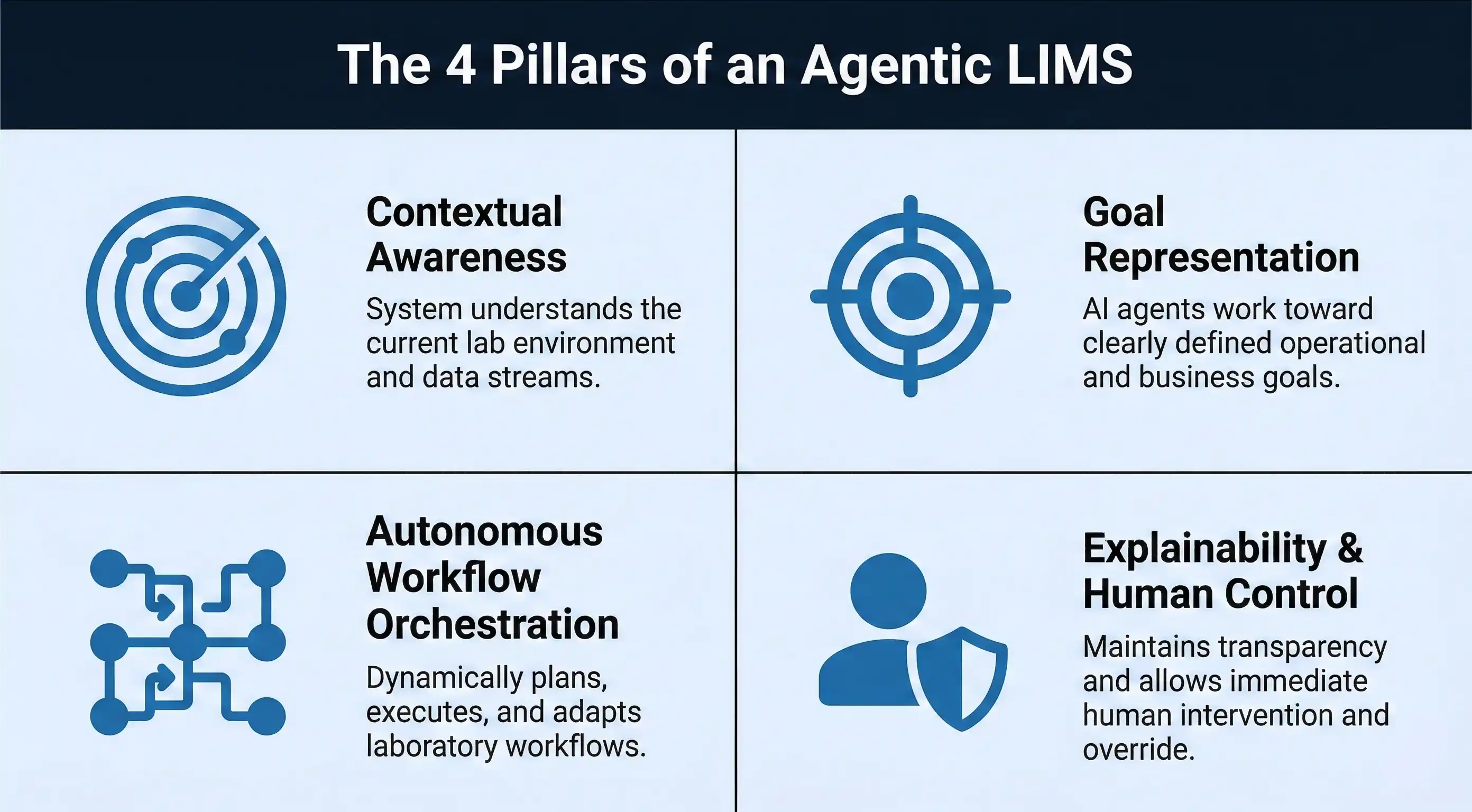
Building a genuinely agentic LIMS requires more than bolting a large language model onto an existing platform. It requires rethinking the system's architecture around four foundational pillars.
Pillar 1: Contextual Awareness
An agentic LIMS must maintain a living, real-time model of the entire laboratory operation. This means continuous integration with instruments (via direct data feeds), ERP systems (for order and client context), environmental monitoring sensors, personnel systems (for shift schedules and competency records), and regulatory calendars. Without rich contextual awareness, an agent cannot make intelligent decisions — it can only make educated guesses.
Pillar 2: Goal Representation
The system must be able to represent and reason about goals at multiple levels simultaneously — strategic goals (maintain ISO 17025 accreditation), operational goals (clear today's sample queue by 5 PM), and immediate tactical goals (resolve this instrument drift before the next calibration check). Goal hierarchies allow the agent to prioritize intelligently when resources are constrained and competing demands arise.
Pillar 3: Autonomous Workflow Orchestration
Rather than running predefined workflows in a fixed sequence, an agentic LIMS dynamically assembles and orchestrates workflows based on current context. It can reroute samples to alternate instruments during downtime, reassign tasks when a technician calls in sick, and compress test sequences intelligently when deadlines tighten — all while maintaining a complete audit trail of every deviation from standard procedure.
Pillar 4: Explainability and Human-in-the-Loop Control
In any regulated environment, 'the AI decided' is never an acceptable explanation for a quality event. Every agentic decision must come with a structured rationale: what information was considered, what alternatives were evaluated, and why this action was selected. Human supervisors must be able to review, override, or approve decisions at configurable checkpoints — and those overrides must themselves be logged as part of the audit trail.
4. How Autonomous LIMS Transforms Laboratory Workflows
The practical impacts of agentic AI in a laboratory context are best understood through the lens of specific workflow scenarios. Consider what changes when a LIMS moves from passive record-keeping to active, goal-directed operation across the five core phases of the sample lifecycle.
| Workflow Phase | Traditional LIMS | Agentic LIMS |
|---|---|---|
| Sample Receipt & Login | Manual data entry; technician assigns tests based on request form. | Agent auto-classifies sample, validates against client specification, assigns optimized test battery, and flags discrepancies — in seconds. |
| Test Scheduling | Supervisor reviews queue and assigns to bench; capacity managed manually. | Agent dynamically schedules tests based on instrument availability, technician competency, sample stability windows, and priority — continuously rebalancing as conditions change. |
| QC Monitoring | SPC alerts notify technician; technician investigates manually. | Agent detects statistical drift, performs root cause pre-analysis, quarantines affected results, initiates corrective action workflow, and escalates with full context — before results are released. |
| Reporting | Analyst compiles results, populates report template, performs manual review. | Agent auto-generates report, cross-references against specification, applies electronic signatures where criteria are met, and delivers to client portal — with a summary of any outliers flagged for analyst review. |
| Compliance & Audit | Audit preparation requires manual data compilation; gaps discovered during audit. | Agent continuously monitors compliance posture, pre-generates audit evidence packages, identifies and closes gaps proactively, and maintains readiness as an ongoing operational state. |
The cumulative effect of these changes is not incremental improvement — it is a structural transformation of what a laboratory can achieve with the same human resources. Labs that historically managed 200 samples per day can operate at 3x capacity. Quality events that took days to investigate get resolved in hours. And the cognitive load on skilled scientists shifts away from administrative coordination toward the analytical work that actually requires human expertise.
5. Agentic AI, Compliance, and the Question of Trust
No conversation about autonomous systems in a regulated laboratory can skip the compliance question. Laboratories operating under ISO 17025, FDA 21 CFR Part 11, GLP, GMP, or CAP accreditation are subject to documentation requirements that leave little room for ambiguity. Every action — human or system-generated — must be traceable to an authorized decision.
This concern is legitimate, and it is one that the best agentic LIMS architectures address by design rather than as an afterthought. The key principles are:
- Full audit trails for agent actions: Every autonomous decision the system makes — rescheduling a test, flagging a result, triggering a corrective action — is logged with a timestamp, the data that informed the decision, the alternatives considered, and the rationale applied. This record is immutable and fully searchable.
- Configurable human approval gates: Laboratories can define which categories of agent action require human approval before execution. Release of results to clients might always require analyst sign-off, while rescheduling within defined parameters might be fully autonomous. The balance is configurable and can be calibrated as trust in the system matures.
- Electronic signature integration: Where 21 CFR Part 11 or equivalent standards apply, agent-initiated workflows include structured e-signature prompts that comply with authentication and non-repudiation requirements.
- Conflict resolution with regulatory precedence: When an agent's optimisation logic conflicts with a regulatory constraint, the constraint wins — always. The system treats compliance as a hard boundary, not a variable to be traded off against efficiency.
🔒 COMPLIANCE BY ARCHITECTURE
At Revol LIMS, we approach regulatory compliance as an architectural property of the system, not a layer of controls applied on top of it. Our agentic AI is being built from the ground up within a framework that treats ISO 17025, 21 CFR Part 11, and GxP requirements as non-negotiable constraints on every autonomous action the system can take.
6. Where Revol LIMS Stands Today
Revol LIMS has been building the foundation for agentic laboratory intelligence for several years — long before the term entered popular usage. Several of the capabilities that define an agentic LIMS architecture are already live and in production across our customer base today.
Our Gen-AI chat assistant allows laboratory staff to interact with the LIMS through natural language — logging samples, querying complex datasets, and generating custom reports through conversational commands. This is the interface through which human goals are communicated to an AI system that then acts on them.
Our predictive analytics engine already operates across the full sample lifecycle — forecasting quality trends, predicting instrument maintenance needs, and anticipating turnaround time risks before they materialise. These are continuously updated, probabilistic models that inform operational decisions in real time.
Our automated anomaly detection monitors QC and test result data streams continuously, flagging deviations from expected patterns at a level of sensitivity that manual review cannot match at scale. Anomalies are not just flagged; they are contextualised — linked to their probable causes, associated with relevant historical events, and routed to the appropriate response workflow automatically.
Our intelligent test prioritisation engine evaluates sample urgency, resource availability, instrument throughput, and contractual commitments simultaneously to produce a continuously optimised work queue — one that adapts in real time as conditions change throughout the operational day.
Taken together, these capabilities represent a platform that is already operating at the frontier of what conventional LIMS can deliver, and that is architected to extend into genuinely agentic territory as the next generation of capabilities comes online.
7. How to Prepare Your Lab for the Agentic Era
Transitioning to an agentic laboratory model is not primarily a technology challenge — it is an operational and organizational one. The technology is advancing rapidly; the harder questions involve how labs structure their data, define their goals, and build the human-AI collaboration models that allow autonomous systems to operate safely within a regulated environment.
Start with data quality. Agentic AI systems are only as intelligent as the data they operate on. Laboratories with fragmented data across disconnected systems, inconsistent naming conventions, or significant volumes of unstructured paper records will find that their data challenges become a constraint on how quickly they can adopt agentic capabilities. The work of data standardization and digitization is foundational and should begin now.
Define what 'good' looks like. Agentic systems need goals to pursue. This means articulating the outcomes your laboratory is trying to optimize — turnaround time, first-pass yield, cost per test, OOS rate, client satisfaction scores — in terms specific enough that an AI system can measure its own progress toward them.
Invest in change management. Laboratory staff who have spent careers developing expertise in managing complex operational situations will reasonably ask how an autonomous LIMS changes their role. The answer is that it elevates it — freeing skilled scientists from coordination overhead and concentrating their work on interpretation, innovation, and exception management.
Choose a platform built to grow. The difference between a LIMS that can grow into agentic capabilities and one that cannot is largely architectural. API-first designs, modular AI frameworks, clean data schemas, and vendor commitment to ongoing R&D are the characteristics to look for when evaluating platforms for the next decade of laboratory operations.
8. Conclusion
The transition from passive record-keeping to active, goal-directed intelligence is the most significant evolution in laboratory information management since the move from paper-based systems to digital platforms. Agentic AI represents a qualitative change in what a LIMS can do — not just storing and surfacing information, but actively working to achieve the laboratory's objectives, continuously, at scale, and with full accountability for every decision it makes.
This is not a distant prospect. The foundations are already in place at laboratories powered by Revol LIMS. The path to a fully agentic laboratory operation is one of progressive capability rollout, each phase building on the last, with human oversight and regulatory compliance maintained at every step.
The laboratories that begin this journey today — investing in data quality, defining operational goals clearly, and building on a platform architected for agentic expansion — will be the ones that define what operational excellence looks like in this industry for the next decade.
The future of laboratory operations is agentic. At Revol LIMS, we are building it.
Ready to Build Your Agentic Laboratory?

Discover how Revol LIMS's AI-powered platform positions your laboratory at the forefront of the agentic era — with compliance built in, implementation in weeks, and a roadmap that grows with you.




















