
Biobank LIMS: Solving Sample Management Challenges & ISO 20387 Compliance
Biobanks are increasingly facing new challenges - not through dramatic errors, but through the slow accumulation of manual gaps: missed freeze-thaw cycles, unlinked aliquots, paper donor logs, and audit trails that exist in three separate systems. Here is how a purpose-configured biobank LIMS closes every one of those gaps.
68%
of biobank sample losses caused by incomplete cold chain documentation
3–7x
cost of reprocessing degraded biospecimens vs. preventing degradation
40%
of biobank audits cite chain-of-custody documentation as primary gap
ISO 20387
international accreditation standard requiring full traceability
WHAT THIS ARTICLE COVERS
- Why Biobank Sample Management Is Inherently Difficult
- The 6 Critical Challenges Biobank LIMS Directly Addresses
- ISO 20387 Compliance and How LIMS Enforces It
- Generic vs. Biobank-Configured LIMS - A Direct Comparison
- What to Look For When Evaluating a Biobank LIMS
- Frequently Asked Questions
1. Why Biobank Sample Management Is Inherently Difficult
A biobank is not simply a cold storage facility that happens to deal with human biological material. It is a precision-custody environment where every decision - from donor intake to aliquot preparation to long-term cryogenic archiving - carries scientific, legal, and ethical weight that few other laboratory disciplines can match.
Biospecimens - blood tubes, serum aliquots, tissue in formalin jars, DNA extracts, PBMC preparations - are irreplaceable. Once degraded, mislabelled, or lost to a broken freeze-thaw record, they cannot be regenerated from a raw material stock. The donor may no longer be contactable. The research window is closed. This is the fundamental asymmetry that defines biobank operations: the cost of a preventable error is always higher than the cost of preventing it.
The challenge is that the workflows required to prevent those errors - complete donor intake documentation, aliquot tracking to parent specimen, freeze-thaw cycle logging, location-level cryogenic inventory, consent status management across a changing regulatory landscape - are genuinely complex. In most biobanks that have grown organically, they are managed across a combination of spreadsheets, paper logs, standalone freezer monitoring systems, and whatever database was available when the operation expanded.
A biobank LIMS configured for biorepository operations does not simply digitise these workflows. It integrates them into a single, auditable environment where every action on every sample is traceable, every compliance checkpoint is enforced at the point of activity, and every deviation from expected parameters triggers an alert before the sample's value is compromised.
2. The 6 Critical Biobank Challenges LIMS Directly Addresses
Challenge 01
Donor Intake and Consent Management Is Disconnected from the Sample Record
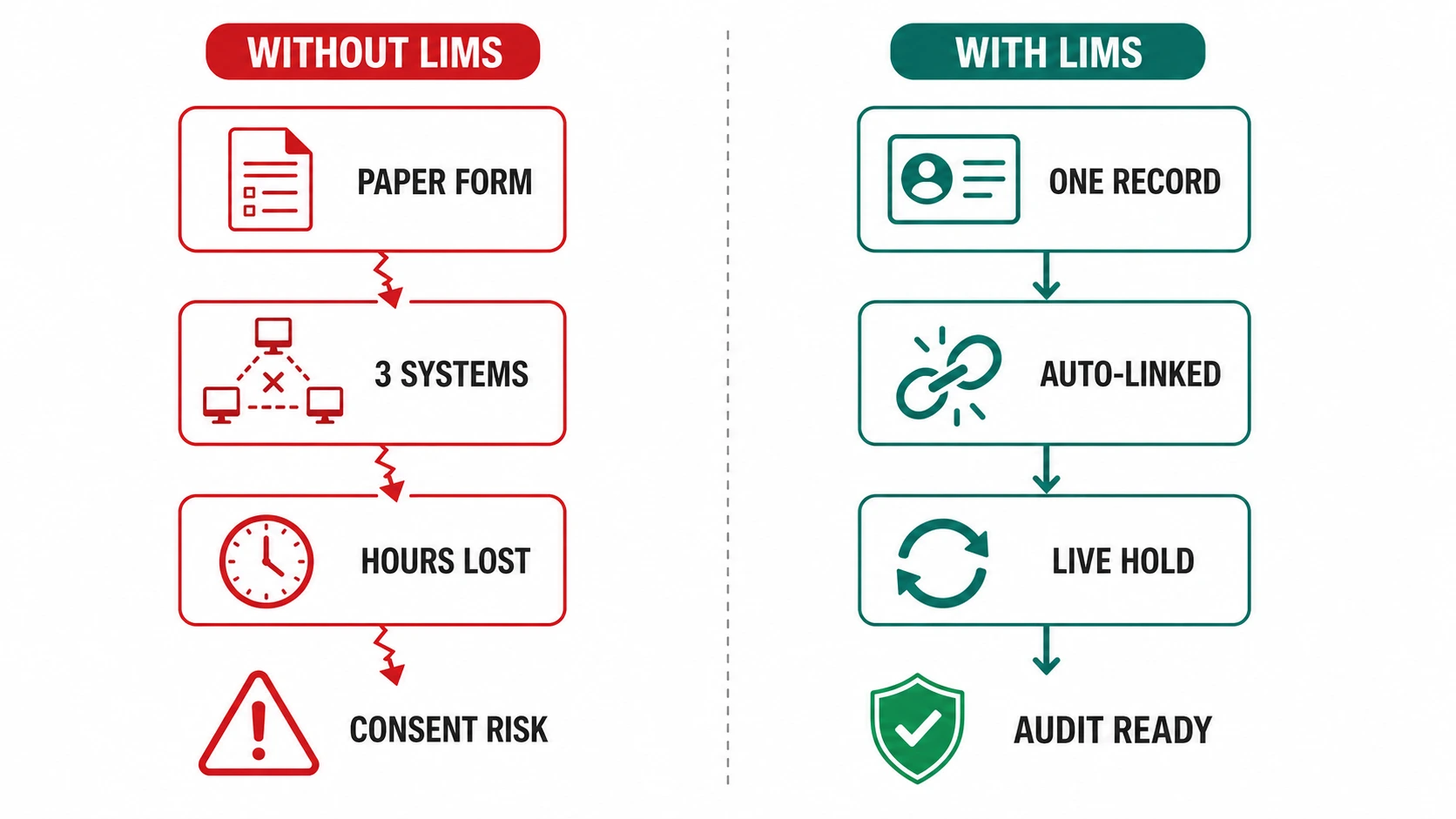
THE CHALLENGE
In most biobanks, donor intake - demographics, medical history flags, consent form execution, ethics protocol reference - is captured in a patient management system or on paper forms that have no live link to the biospecimen record. When a research request comes in, staff must cross-reference multiple systems to confirm consenting donors have authorised the proposed use - a process that takes hours and introduces transcription risk at every step.
HOW BIOBANK LIMS FIXES IT
- Donor intake forms captured directly in the biobank LIMS at collection, creating a single record linking demographics, consent type, ethics reference, and all derived specimens.
- Consent scope enforced at the sample request stage - LIMS prevents allocation to any research use not covered by the consented purpose.
- Consent withdrawal events update the donor record in real time, placing all associated specimens on a restricted-use hold across the inventory.
- Audit-ready consent reports generated per donor, per collection event, or per ethics protocol without manual data assembly.
Challenge 02
Aliquot Tracking Has No Reliable Parent-Child Relationship
THE CHALLENGE
A primary blood draw may produce a serum tube, three EDTA aliquots, a cell pellet preparation, and a residual archival fraction - all derived from the same collection event but potentially processed by different staff, stored in different freezers, and entered as independent records with no formal link. When downstream integrity verification or volume reconciliation is required, the link must be reconstructed manually - if it can be reconstructed at all.
HOW BIOBANK LIMS FIXES IT
- Every aliquotting event creates a documented parent-child relationship - derived specimens automatically linked to the primary collection record.
- The primary specimen record shows a full derivative tree: every aliquot, its current location, volume, usage history, and freeze-thaw count.
- Sample splitting operations tracked with user authentication, timestamp, and volume reconciliation - discrepancies flagged immediately.
Challenge 03
Freeze-Thaw Cycle Logging Is Manual and Inconsistently Applied
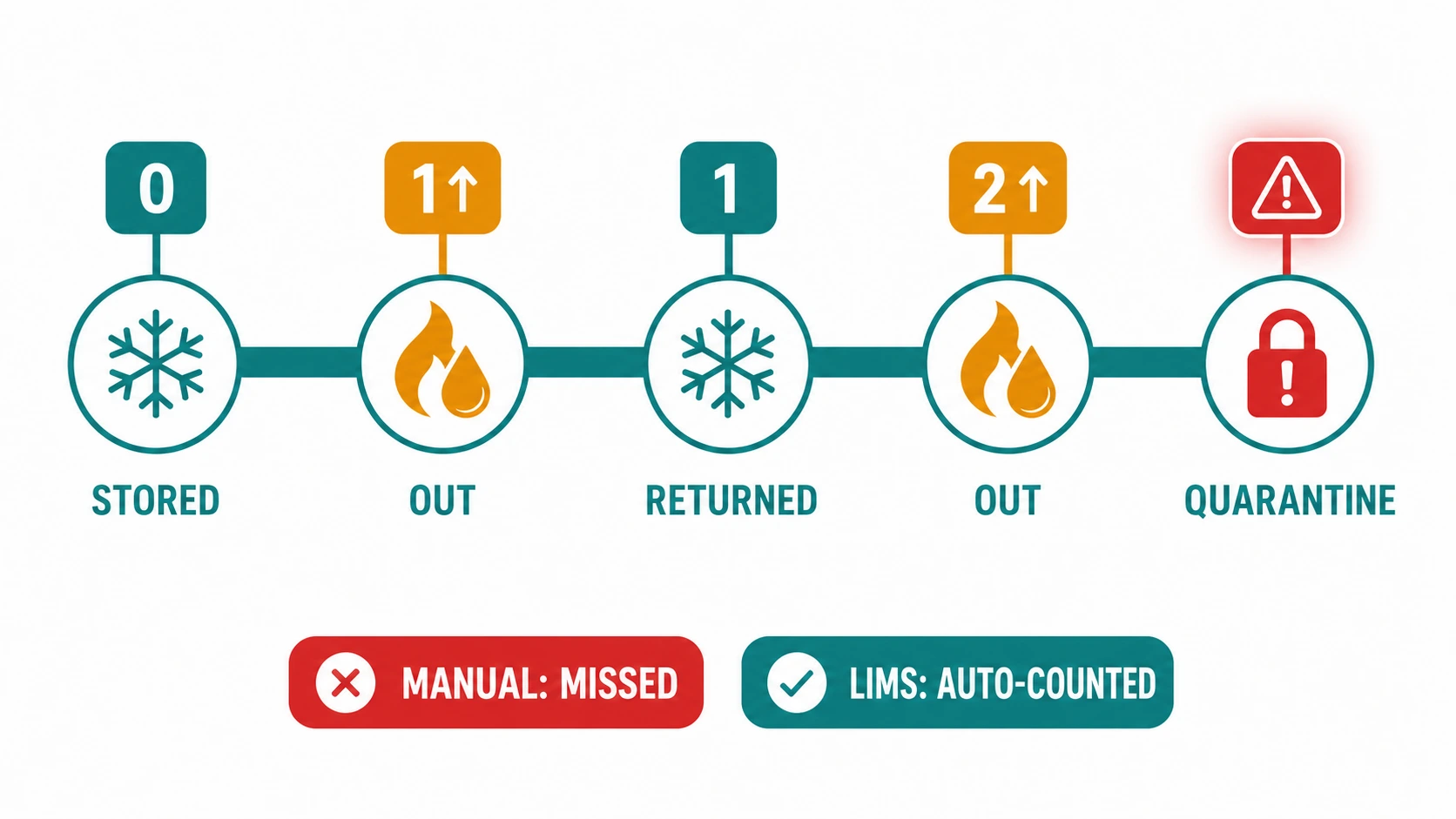
THE CHALLENGE
Freeze-thaw cycles are one of the most significant determinants of biospecimen quality, particularly for protein-sensitive samples, RNA preparations, and DNA aliquots. Every uncontrolled thaw degrades the sample's scientific utility. In most biobanks, freeze-thaw events are only recorded when staff remember to record them — often a tally mark on a paper label. A researcher requesting a sample may have no reliable way of knowing how many times it has been frozen and thawed.
HOW BIOBANK LIMS FIXES IT
- Every retrieval from cryogenic storage requires an authenticated LIMS check-out, automatically incrementing the freeze-thaw counter on the specimen record.
- Every retrieval from cryogenic storage requires an authenticated LIMS check-out, automatically incrementing the freeze-thaw counter on the specimen record.
- Thresholds for acceptable freeze-thaw counts set per sample type - samples exceeding the threshold are flagged and quarantined from research allocation.
- Freeze-thaw history included in every sample dispatch record provided to requesting researchers, meeting ISO 20387 disclosure requirements.
Challenge 04
Cryogenic Inventory Management Depends on Physical Inspection
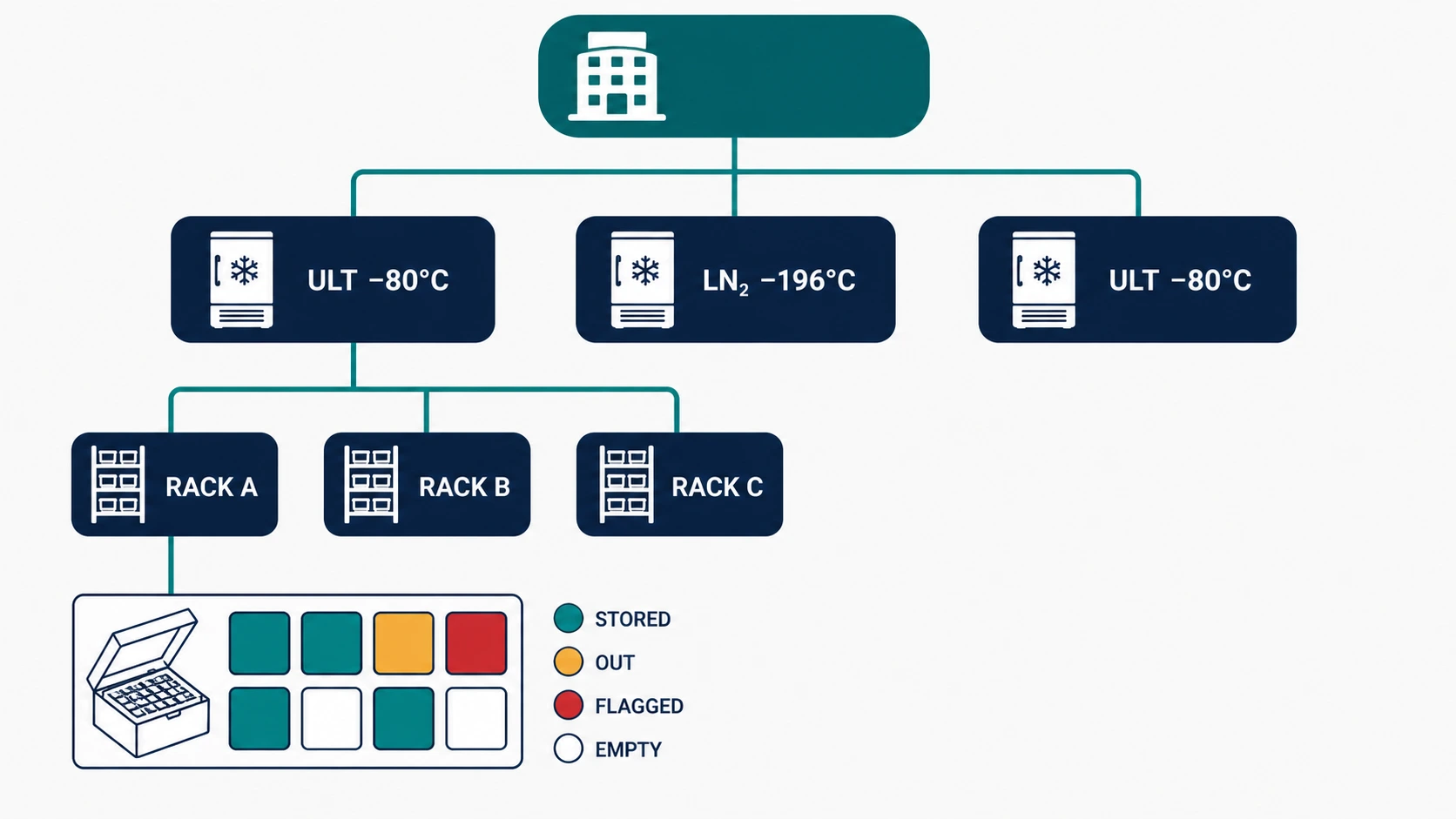
THE CHALLENGE
A biobank with 50,000 archived specimens across multiple ULT freezers and liquid nitrogen tanks has a fundamental logistics challenge: knowing what is where in real time. Location drift - where the system record and physical reality diverge - is one of the most common operational problems in biobanks and one of the most time-consuming to diagnose and correct.
HOW BIOBANK LIMS FIXES IT
- Position-level inventory tracks every specimen to a specific freezer - shelf - rack - box - well position, updated at every movement event.
- Barcode or RFID scan at check-out, and check-in enforces location accuracy without relying on manual data entry.
- Freezer capacity dashboards show current occupancy per unit, supporting space planning and pre-empting storage crises.
- Equipment downtime events - freezer failure, planned maintenance - trigger automatic inventory alerts for all specimens at risk.
Challenge 05
Chain of Custody Gaps Create Undefendable Sample Records
THE CHALLENGE
A biospecimen used in published research carries an implicit assertion: that the sample was what it was claimed to be, collected when and from whom the record states, stored under the conditions described, and handled only by authorised personnel for authorised purposes. If the chain of custody has gaps, that assertion cannot be fully supported in a regulatory audit, research integrity review, or legal proceeding involving donor consent.
HOW BIOBANK LIMS FIXES IT
- Every sample handling event - receipt, aliquotting, storage, retrieval, dispatch - requires an authenticated user action creating an immutable, timestamped audit entry.
- The LIMS enforces a chain of custody at every transfer point, preventing sample movement without a recorded custody handoff between named users.
- Deviations - temperature excursion, unexpected access, delayed processing - logged automatically and flagged for QA review, not silently absorbed.
- Complete chain of custody reports generated on demand for any specimen, formatted for regulatory submission or research publication.
Challenge 06
Research Request Fulfilment Is a Manual, Error-Prone Process
THE CHALLENGE
When a researcher submits a request for samples - specimens meeting specific clinical criteria, consented for the proposed use, with freeze-thaw cycles remaining - the biobank manager manually queries inventory, cross-references the consent database, checks against active allocations, and assembles the pick list. This scales poorly, and errors lead to over-allocation and consent mismatches.
HOW BIOBANK LIMS FIXES IT
- Research request workflows managed natively in LIMS - structured request forms automatically query the inventory against all selection criteria.
- Allocation logic prevents double-booking - once committed to a request, a sample is flagged as reserved across all concurrent searches.
- Consent enforcement applied automatically at the point of allocation - no manual cross-referencing against a separate consent register.
- Dispatch workflows generate chain of custody documentation, shipping manifests, and receipt confirmation templates, all linked to the originating request record.
3. ISO 20387 Compliance and How Biobank LIMS Enforces It
ISO 20387 is the international standard for the competence, impartiality, and consistent operation of biobanks. Published in 2018 and increasingly required by funding bodies, ethics committees, and collaborative research networks, it sets explicit requirements across pre-analytical sample handling, documentation integrity, quality management, and long-term storage conditions.
The standard does not mandate a specific technology. But its documentation requirements - traceability at every processing step, audit-ready records of storage conditions, documented and controlled deviation handling, version-controlled SOPs accessible at the point of activity - are practically impossible to meet at scale without an integrated biobank LIMS.
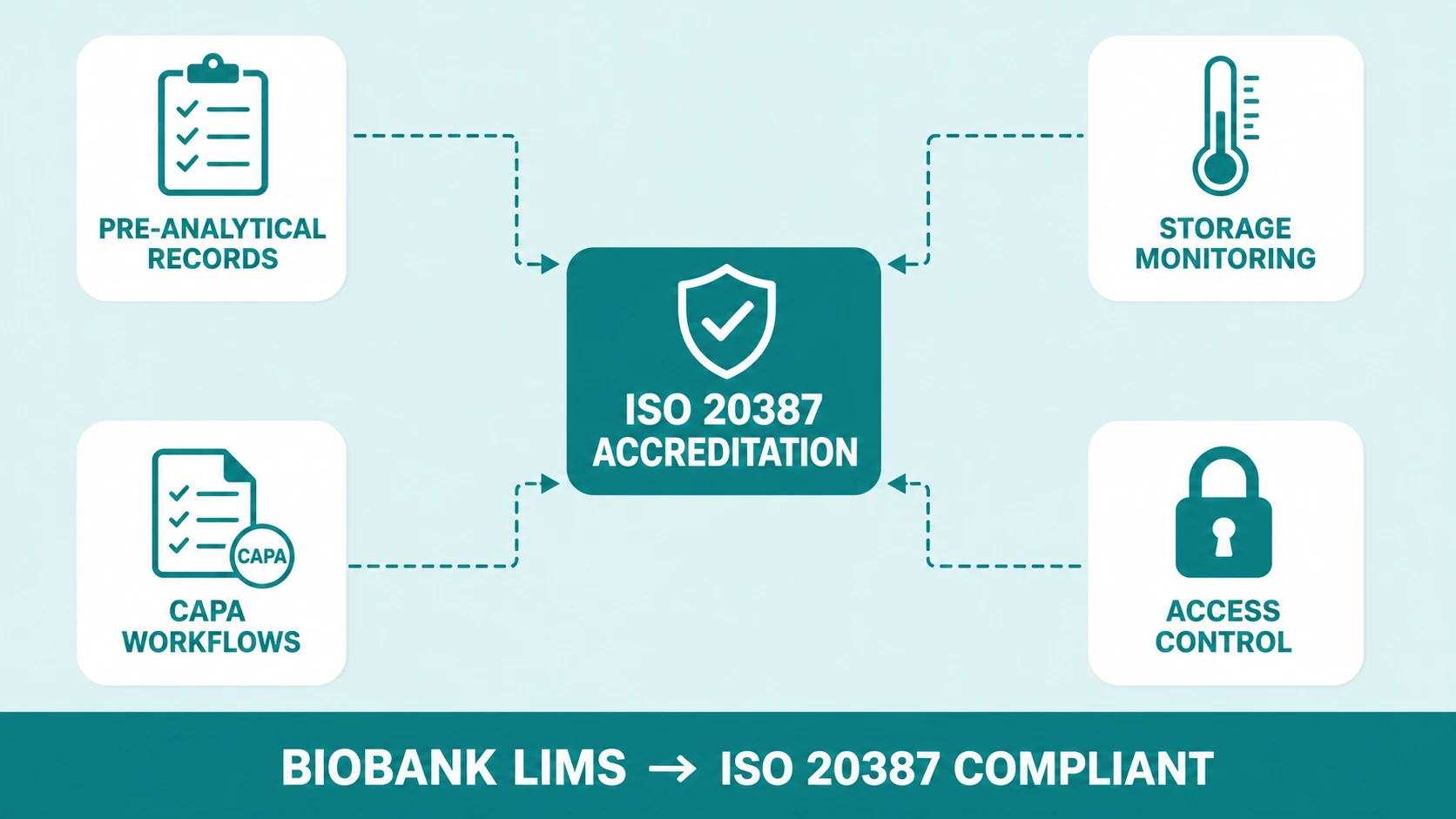
Pre-analytical process documentation
The standard requires that all pre-analytical variables — collection time, transport conditions, processing delay, centrifugation parameters — be recorded for every specimen. A biobank LIMS captures these as structured fields at intake. Deviations from defined SOP parameters are flagged at the point of entry, not discovered during an audit weeks later.
Storage condition monitoring
ISO 20387 requires continuous monitoring of storage conditions. A LIMS integrated with environmental monitoring equipment logs temperature and humidity data against the inventory, linking storage excursion events to the specific specimens at risk — not just a general freezer alarm log that requires manual interpretation.
Quality management and CAPA
Non-conformance events — sample degradation, mislabelling, consent issues, equipment failures — must be documented, investigated, and resolved through a structured CAPA process. A biobank LIMS provides the workflow to log non-conformances, assign corrective actions, track completion, and link the closed CAPA record to the affected specimens.
Impartiality and access control
ISO 20387 requires that access to biospecimens and related data be controlled and documented. A biobank LIMS enforces role-based access at the record level — every access event is logged automatically, without relying on staff to remember to record it.
4. Generic vs. Biobank-Configured LIMS — A Direct Comparison
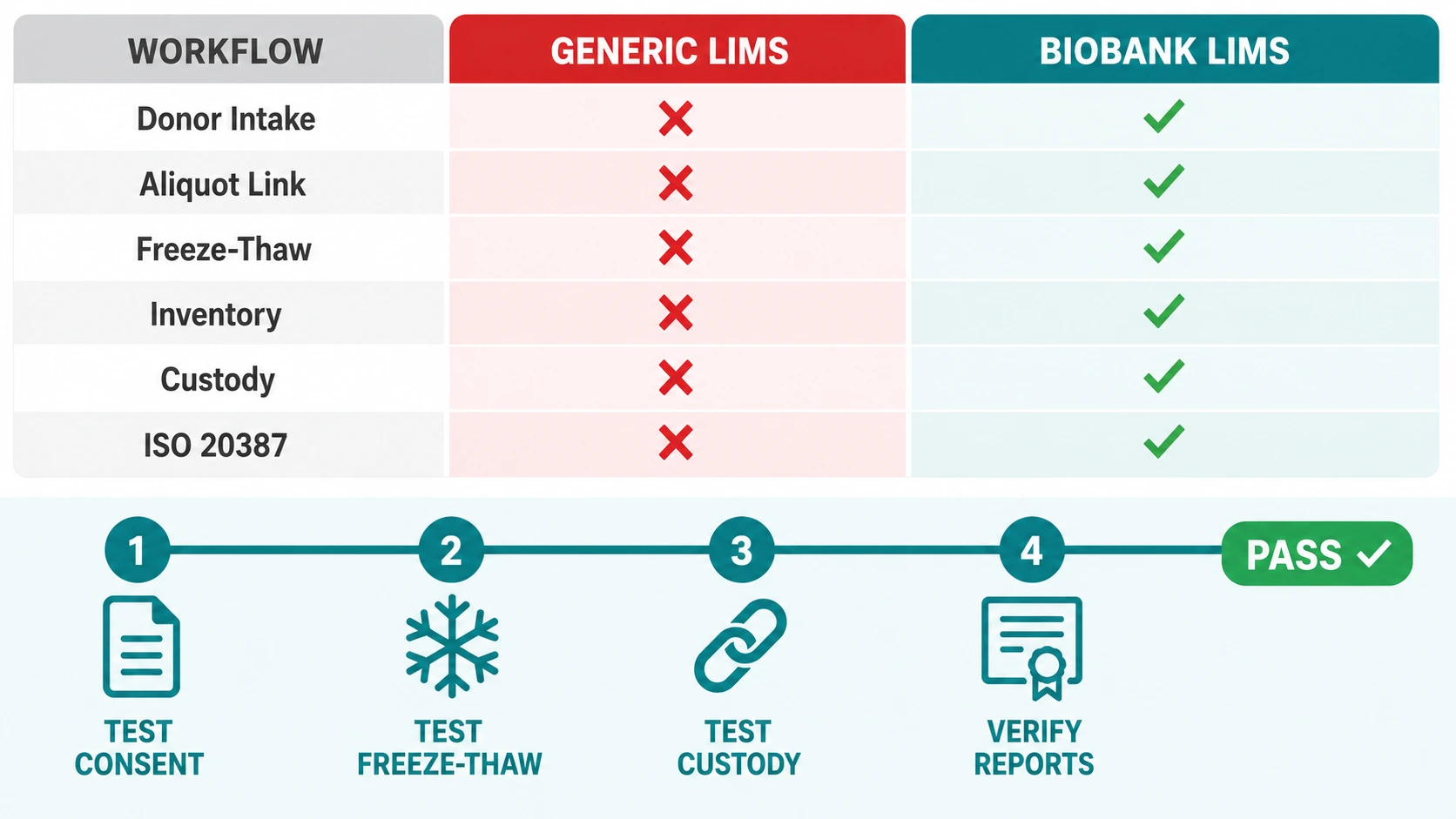
| Workflow Area | Generic LIMS | Biobank-Configured LIMS |
|---|---|---|
| Donor Intake | Sample registered only. Donor data in separate system. | Demographics + consent + ethics captured at intake. Single linked record. |
| Aliquot Tracking | Independent records. Parent-child relationship not enforced. | Every derivative linked to primary specimen. Full family tree queryable. |
| Freeze-Thaw | Manual entry. No automated increment. Inconsistently applied. | Auto-incremented at authenticated check-out. Threshold alerts per sample type. |
| Inventory Location | Box-level only. Location drift common. | Position-level tracking. Barcode/RFID enforced. Real-time dashboards. |
| Chain of Custody | Receipt and result logged. Intervening transfers undocumented. | Every handling event authenticated. Gaps are system-prevented. |
| Consent Enforcement | Not integrated. Manual cross-reference required. | Enforced at allocation. Withdrawal events auto-restrict specimens. |
| Research Fulfilment | Manual query. Double-booking risk. No audit trail. | Structured workflow. Allocation lock. Dispatch generates custody docs. |
| ISO 20387 Readiness | Partial. Manual supplements required. | Native. Audit-ready reports on demand. CAPA workflows built in. |
5. What to Look For When Evaluating a Biobank LIMS
Ask about consent management depth
Request a demonstration of what happens when a donor withdraws consent after samples have been archived and partially allocated to research projects. A genuine biobank LIMS should show automatic restriction of all associated specimens, notification to relevant project holders, and a quarantine workflow — not a note field that staff must remember to check.
Test the freeze-thaw tracking with a realistic scenario
Ask the vendor to simulate a sample retrieved twice for QC testing and once for a failed research allocation before being archived again. The system should show a freeze-thaw count of three, automatically derived from check-out and check-in events, without manual entry.
Probe the chain of custody enforcement model
Ask explicitly: Can a sample be physically moved from one storage unit to another without a LIMS action being taken? If yes, the chain of custody is dependent on staff discipline, not system design. Enforcement means structurally preventing uncustodied movement.
Verify ISO 20387 report generation
Ask to see a pre-analytical quality report, a storage condition compliance summary, and a complete chain of custody document for a sample from intake to dispatch. These are not reports a vendor should need to build for a demo — they should be standard outputs in any biobank-ready LIMS.
6. Frequently Asked Questions
Q1. Can a generic clinical LIMS be configured to meet biobank requirements?
Some enterprise platforms offer sufficient configurability — but the gap between a standard clinical LIMS and a purpose-built biorepository LIMS is substantial. Capabilities most difficult to retrofit include position-level inventory, consent-scope enforcement at allocation, and automated freeze-thaw counting tied to physical check-out events.
Ask any vendor: is another ISO 20387-compliant biobank currently running this configuration in production?
Q2. What is the difference between chain-of-custody tracking and enforcement?
Tracking records what happened — it logs custody events and timestamps them. Enforcement prevents uncustodied movement — it makes it structurally impossible to move a sample without a system-recorded transfer. ISO 20387 requires that the integrity of the custody record be guaranteed, not merely that custody events be recorded when staff remember to log them.
Q3. How does a biobank LIMS handle consent withdrawal under GDPR?
A purpose-configured biobank LIMS handles consent withdrawal as a first-class workflow event: automatically restricting all associated specimens, identifying active research allocations, notifying project managers, and generating a withdrawal action report. Under GDPR's right to erasure, the LIMS must also dissociate donor-identifying data from specimen records while preserving non-personal quality and processing data,the biobank may legitimately retain.
Q4. What are the LIMS validation requirements for ISO 20387 accreditation?
Validation scope should encompass every biobank-specific workflow: donor intake, aliquot creation, freeze-thaw tracking, location management, chain of custody enforcement, consent management, CAPA, and report generation. IQ, OQ, and PQ protocols should use biobank-specific test scripts. Site-specific configuration always requires PQ testing beyond the vendor's standard package.
Q5. How does biobank LIMS help manage sample quality across long-term cryogenic archives?
On the prevention side: automated freeze-thaw monitoring, temperature excursion alerts tied to specific specimens, and equipment status tracking. On the documentation side: every quality-relevant event — processing delay, deviation, equipment fault — is linked to affected specimens. When a researcher requests a sample involved in a temperature excursion year earlier, that information is in the LIMS record and disclosed at the point of allocation.
Request a Demo: https://revollims.com/lims-demo
Explore Biorepository LIMS: https://revollims.com/lims-software/biorepository-lims
Final Thoughts
Biobanks carry an unusual kind of accountability. The samples they manage are not raw materials — they are irreplaceable fragments of human biology, donated for a purpose, stored under conditions that must be maintained, and used in ways that must be authorised. Every gap in the management of those samples — every undocumented freeze-thaw cycle, every consent record that doesn't travel with the specimen, every location entry made three days after the fact — is a gap in the scientific and ethical foundation of the research they support.
A purpose-configured biobank LIMS does not merely reduce administrative burden. It makes the biobank's claims about its specimens defensible — to auditors, to ethics committees, to collaborating researchers, and ultimately to the donors whose consent makes the entire operation possible. That is not a software feature. It is the operational standard the discipline requires.
CONTINUE READING
- Toxicology Lab Workflows: Why Generic LIMS Implementations Fail - https://revollims.com/toxicology-lims-implementation-failures
- Achieving ISO/IEC 17025 Compliance: How a Modern LIMS Simplifies Accreditation https://revollims.com/iso-17025-compliance-modern-lims
- How to Prepare Your LIMS for an FDA Audit: A Practical Checklist - https://revollims.com/lims-fda-audit-checklist
- Revol Biorepository LIMS Feature Overview -
https://revollims.com/lims-software/biorepository-lims
Author: Revol LIMS Team




















